(A) The interior electron cloud (light blue) shields the outer electron of interest from the full attractive force of the nucleus. The Z eff experienced by an electron in a given orbital depends not only on the spatial distribution of the electron in that orbital but also on the distribution of all the other electrons present. Consequently, the Z eff is always less than the actual nuclear charge, Z. An effective nuclear charge is the nuclear charge an electron actually experiences because of shielding from other electrons closer to the nucleus ( Figure 3). As a result, the electron farther away experiences an effective nuclear charge ( Z eff). Hence these electrons will cancel a portion of the positive charge of the nucleus and thereby decrease the attractive interaction between the nucleus and the electron farther away. 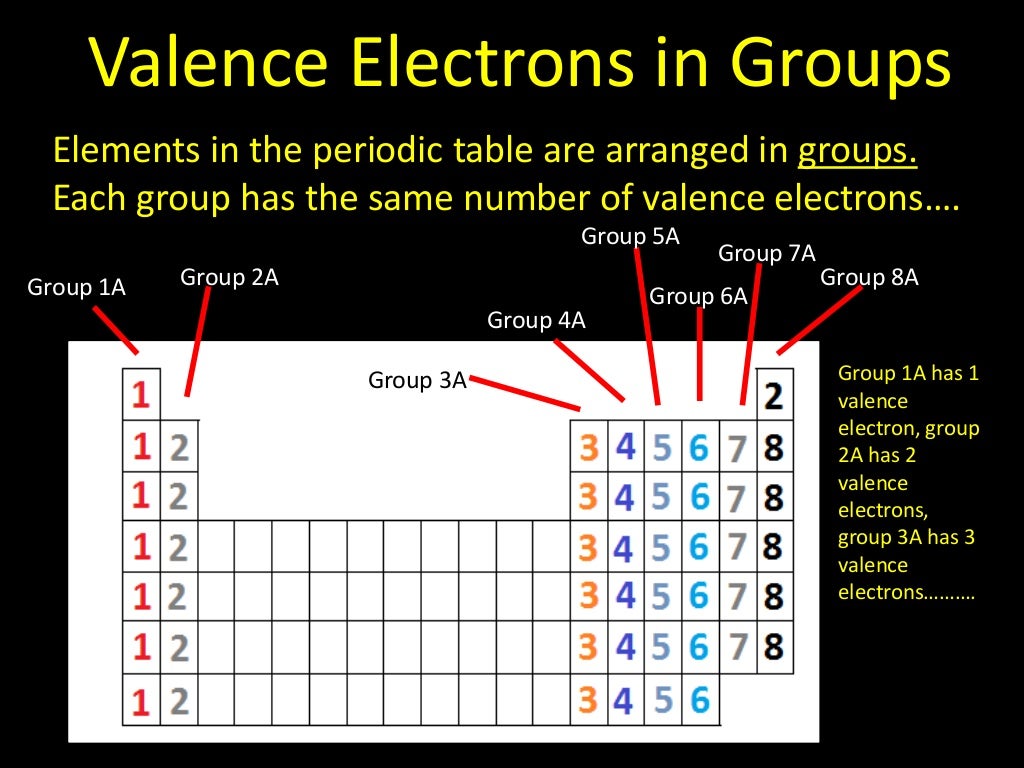
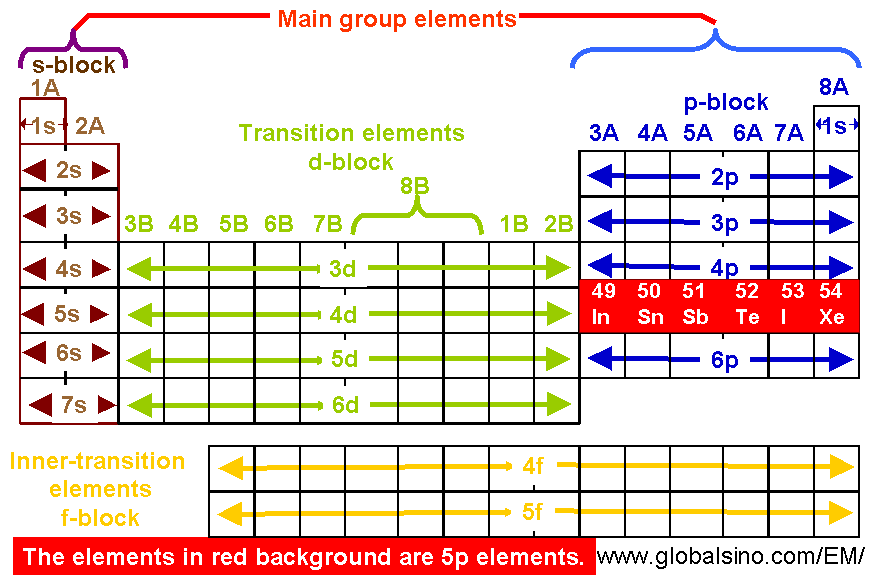
If an electron is far from the nucleus (i.e., if the distance r between the nucleus and the electron is large), then at any given moment, most of the other electrons will be between that electron and the nucleus. There are no known solutions to the Schrodinger equation for this problem, so one must use approximate methods to find the orbitals and their energies. In addition there are attractive interactions between each of the two electrons with the nucleus. From Coloumb’s law we know that there is a repulsive interaction that depends on the distance between them. For example, in helium there are two electrons. When more than one electron is present, however, the total energy of the atom or the ion depends not only on attractive electron-nucleus interactions but also on repulsive electron-electron interactions. Effective Nuclear Charge (Z eff)įor an atom or an ion with only a single electron, we can calculate the potential energy of an electron by considering only the electrostatic attraction between the positively charged nucleus and the negatively charged electron. Note that down each group, the configuration is often similar. This version of the periodic table shows the outer-shell electron configuration of each element. Figure 2 shows the ground-state electron configuration for the valence electrons of these elements as well as that for atoms of each of the known elements. The electron configurations of silicon (14 electrons), phosphorus (15 electrons), sulfur (16 electrons), chlorine (17 electrons), and argon (18 electrons) are analogous in the electron configurations of their outer shells to their corresponding family members carbon, nitrogen, oxygen, fluorine, and neon, respectively, except that the principal quantum number of the outer shell of the heavier elements has increased by one to n = 3. All three atoms have a filled s subshell outside their filled inner shells.Īluminum (atomic number 13), with 13 electrons and the electron configuration 3 s 23 p 1, is analogous to its family member boron, 2 s 22 p 1. The alkaline earth metal magnesium with its 12 electrons in a 3 s 2 configuration, is analogous to its family members beryllium, 2 s 2, and calcium, 4 s 2. All three atoms, which are in the alkali metal family, have only one electron in a valence s subshell outside a filled set of inner shells. Writing the configurations in this way emphasizes the similarity of the configurations of lithium, sodium and potassium. 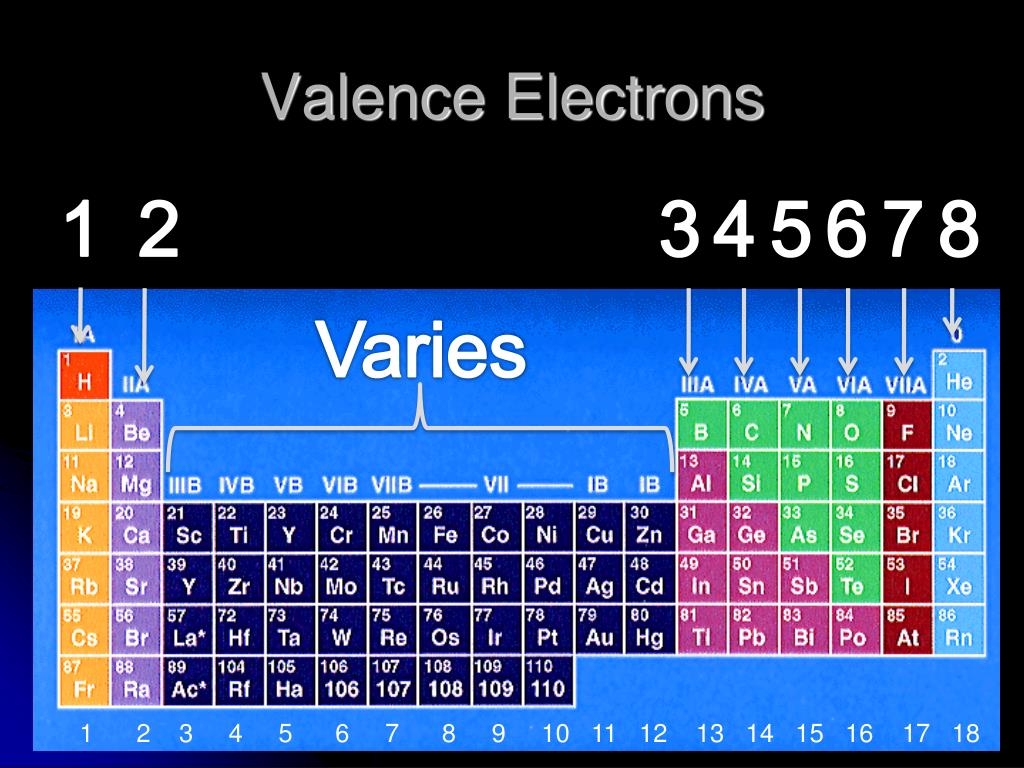
And the abbreviated configuration of potassium, 4 s 1, where represents the configuration of the argon atom, which is identical to that of the filled inner shell of potassium. A core-abbreviated electron configuration (right) replaces the core electrons with the noble gas symbol whose configuration matches the core electron configuration of the other element.Ĭompare this to the abbreviated configuration of lithium, 2 s 1, where represents the configuration of the helium atom, which is identical to that of the filled inner shell of lithium. For sodium, 3 s 1, the symbol represents core electrons (1 s 22 s 22 p 6) and the valence electron is the electron in the 3 s orbital. For some examples, let us consider the ground state electron configurations of the elements in the 3rd period of the periodic table. They can be more easily distinguished when using the noble gas configuration. The electrons occupying the outermost shell orbital(s) are called valence electrons, and those occupying the inner shell orbitals are called core electrons ( Figure 1). | Key Concepts and Summary | Key Equations | Glossary | End of Section Exercises | Core and Valence Electrons 
| Effective Nuclear Charge (Z eff) | Shielding | Correlate the effective nuclear charge with selected trends in periodic properties.Distinguish between core and valence electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
